Leather Shoe Sample Validation: Complete B2B Quality Guide for Manufacturing Excellence
Sample validation represents the critical gateway between design concept and mass production in leather shoe manufacturing. This comprehensive quality control process determines whether your footwear meets specifications, regulatory standards, and market expectations before committing to full production runs. A robust validation framework can reduce defect rates from industry averages of 2-4% to under 1.2%, while ensuring consistent quality across thousands of pairs.
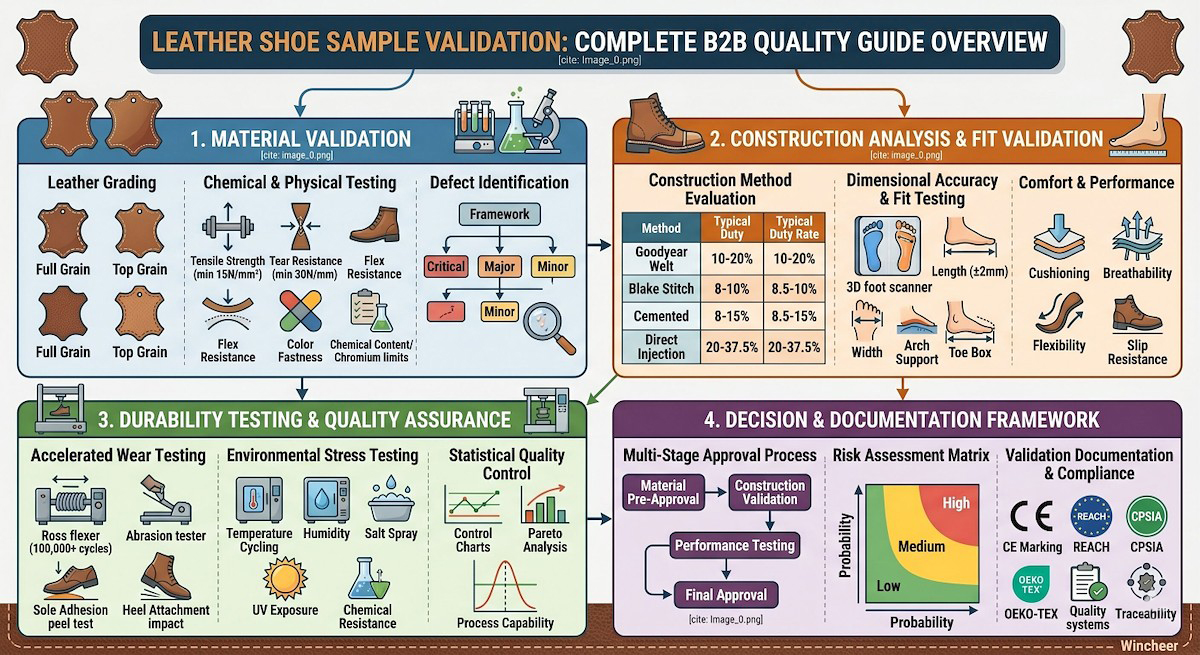
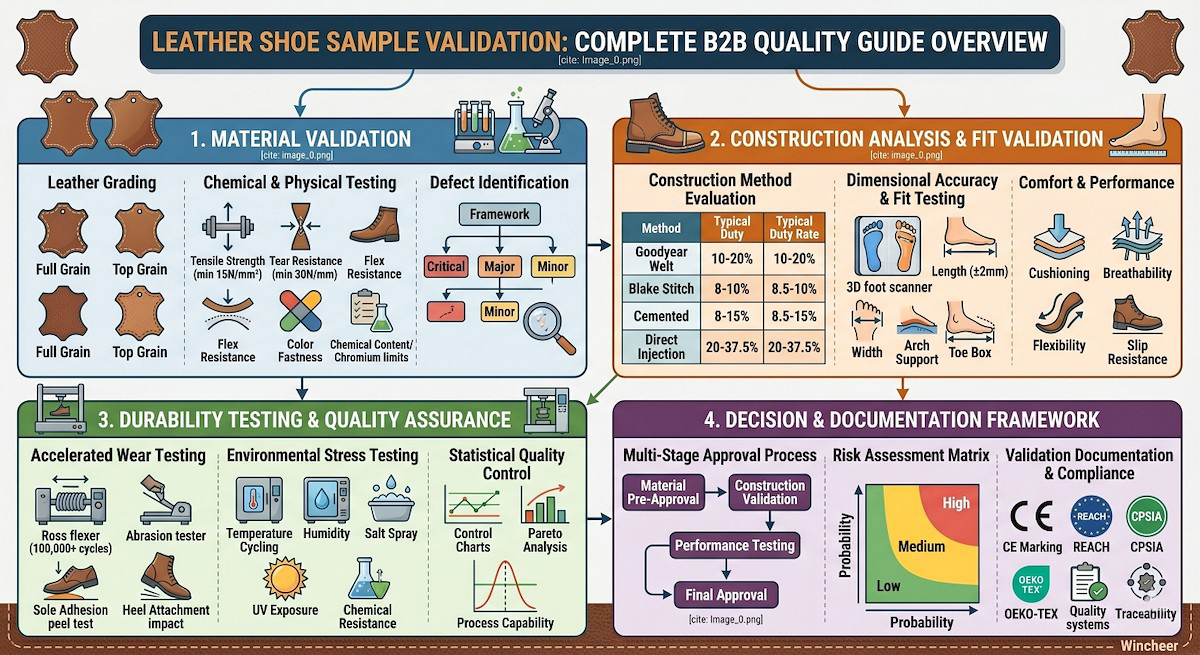
The leather shoe sample validation process encompasses material verification, construction analysis, fit testing, durability assessment, and compliance certification. Each validation stage serves as a quality checkpoint, preventing costly production errors and customer returns. Understanding this process empowers B2B buyers, private label brands, and cross-border sellers to make informed sourcing decisions and maintain quality standards that protect their brand reputation.
In our factory at Wincheer, established in 2007, Shoes, we’ve refined our sample validation protocols over 17 years of manufacturing excellence, producing 500,000+ pairs annually with defect rates consistently below 1.2%. Our comprehensive validation process covers everything from leather grading to final quality inspection, ensuring each design meets CE (EN ISO20347), REACH, and RoHS certifications before approval. This systematic approach has enabled us to serve 200+ partners across 50+ countries while maintaining zero MOQ on existing designs and just 60 pairs for custom developments.
Leather Shoe Sample Validation: Understanding Leather Quality Standards and Material Validation
Leather Grade Classification Systems
Leather quality forms the foundation of sample validation, requiring systematic assessment of hide characteristics, processing quality, and performance attributes. Professional leather grading follows established industry standards that evaluate grain structure, thickness consistency, chemical treatment, and surface finish quality.
Primary Leather Grades:
- Full Grain Leather: Highest quality with intact grain surface, maximum durability
- Top Grain Leather: Sanded surface, consistent appearance, good durability
- Genuine Leather: Split leather with artificial grain, budget-friendly option
- Bonded Leather: Reconstituted leather fibers, lowest quality grade
Material validation begins with visual inspection under controlled lighting conditions, checking for grain consistency, color uniformity, and surface defects. Thickness measurements using precision gauges ensure compliance with specifications, typically ranging from 1.2-2.0mm for dress shoes and 1.8-2.5mm for boots.
Chemical and Physical Testing Protocols
Comprehensive leather testing validates chemical composition, physical properties, and performance characteristics. https://www.iso.org/standard/70740.html – SATRA Footwear Technology ISO standards provide frameworks for leather testing methodologies, ensuring consistent evaluation criteria across manufacturers.
Essential Testing Parameters:
- Tensile Strength: Measures leather’s resistance to tearing (minimum 15 N/mm²)
- Tear Resistance: Evaluates durability under stress (minimum 30 N/mm)
- Flex Resistance: Tests cracking resistance through repeated bending cycles
- Color Fastness: Assesses color stability against light, water, and friction
- Chemical Content: Verifies chromium levels, formaldehyde content, and banned substances
Testing equipment includes tensile testing machines, flexometers, color matching cabinets, and chemical analysis instruments. Results must meet regulatory requirements including REACH compliance for European markets and CPSIA standards for children’s footwear.
Leather Defect Identification Framework
Systematic defect identification prevents substandard materials from entering production. Common leather defects include grain breaks, scars, insect bites, stains, and processing irregularities that compromise appearance or durability.
Defect Classification System:
- Critical Defects: Structural damage affecting durability or safety
- Major Defects: Appearance issues impacting marketability
- Minor Defects: Cosmetic flaws within acceptable tolerance ranges
Documentation protocols require photographic evidence, defect location mapping, and severity assessment scores. This data informs supplier feedback and continuous improvement initiatives.
Construction Analysis and Fit Validation Procedures
Shoe Construction Method Evaluation
Construction method validation ensures manufacturing techniques align with design specifications and quality requirements. Different construction methods offer varying durability, comfort, and aesthetic characteristics that impact final product performance.
| Construction Method | Durability Rating | Water Resistance | Resoling Capability | Typical Price Range |
|---|---|---|---|---|
| Goodyear Welt | Excellent (8-10 years) | Good | Yes | $80-200+ |
| Blake Stitch | Good (5-7 years) | Moderate | Yes | $60-150 |
| Cemented | Fair (2-4 years) | Poor | No | $25-80 |
| Direct Injection | Good (4-6 years) | Excellent | No | $40-100 |
Construction validation examines stitch quality, adhesive application, sole attachment strength, and component alignment. Tensile testing of sole-to-upper bonds ensures adequate strength for intended use conditions.
Dimensional Accuracy and Fit Testing
Fit validation requires systematic measurement of internal dimensions, last accuracy, and size consistency across production samples. Digital foot scanners and 3D measurement systems provide precise dimensional data for comparison against target specifications.
Critical Fit Parameters:
- Length Accuracy: ±2mm tolerance from size standards
- Width Consistency: Proper last selection for target demographics
- Arch Support: Anatomical alignment with foot structure
- Heel Cup Depth: Secure heel retention without pressure points
- Toe Box Volume: Adequate space for natural toe movement
Fit testing protocols involve multiple foot shapes and sizes, documenting pressure points, comfort levels, and sizing recommendations. This data informs size grading and last modifications before production approval.
Comfort and Performance Validation
Comfort validation extends beyond dimensional accuracy to evaluate cushioning, breathability, flexibility, and support characteristics. Standardized testing protocols measure these attributes objectively, supplemented by wearer trials for subjective feedback.
Performance Testing Methods:
- Cushioning Analysis: Impact absorption measurement using drop tests
- Breathability Testing: Air permeability and moisture vapor transmission rates
- Flexibility Assessment: Bend resistance and recovery characteristics
- Slip Resistance: Coefficient of friction on various surfaces
- Temperature Performance: Thermal insulation and heat dissipation properties
Results comparison against established benchmarks determines whether samples meet performance targets for intended market segments and use cases.
Durability Testing and Quality Assurance Protocols
Accelerated Wear Testing Procedures
Durability validation employs accelerated testing methods that simulate extended wear periods in controlled laboratory conditions. These tests predict product lifespan and identify potential failure modes before market introduction.
Standard Durability Tests:
- Flexing Resistance: 100,000+ flex cycles simulating walking motion
- Abrasion Testing: Surface wear resistance under controlled conditions
- Sole Adhesion: Peel strength testing of sole-to-upper bonds
- Heel Attachment: Impact and fatigue testing of heel construction
- Hardware Durability: Corrosion resistance and mechanical strength of buckles, eyelets
Testing equipment includes Ross flexing machines, Martindale abrasion testers, and universal testing machines. Results interpretation requires understanding of correlation factors between laboratory conditions and real-world wear patterns.
Environmental Stress Testing
Environmental validation exposes samples to temperature extremes, humidity variations, and chemical exposure that products may encounter during storage, shipping, and use. This testing reveals material degradation patterns and construction vulnerabilities.
Environmental Test Conditions:
- Temperature Cycling: -20°C to +60°C exposure cycles
- Humidity Testing: 95% RH at elevated temperatures
- Salt Spray: Corrosion resistance for hardware components
- UV Exposure: Color fastness and material degradation
- Chemical Resistance: Exposure to common cleaning agents and solvents
Documentation includes before/after photographs, dimensional changes, and performance parameter variations. Pass/fail criteria align with intended market requirements and regulatory standards.
Statistical Quality Control Implementation
Quality control validation employs statistical methods to ensure sample representativeness and production consistency. Control charts, process capability studies, and acceptance sampling plans provide objective quality assessment frameworks.
[Shoe Manufacturer Guide](https://the manufacturer.com/shoe-manufacturer/)
Statistical Tools:
- Control Charts: Track key quality parameters over time
- Process Capability Analysis: Assess manufacturing consistency
- Acceptance Sampling: Determine inspection sample sizes and criteria
- Defect Pareto Analysis: Identify primary quality issues for improvement focus
Implementation requires trained quality personnel, calibrated measurement equipment, and documented procedures that ensure consistent application across all validation activities.
Sample Validation Decision Framework
Multi-Stage Approval Process
Effective sample validation follows a structured decision framework that progresses through defined stages, each with specific approval criteria and documentation requirements. This systematic approach prevents subjective decisions and ensures consistent quality standards.
Validation Stage Framework:
Stage 1: Material Pre-Approval
- Leather quality assessment
- Component specification verification
- Supplier certification review
- Initial cost analysis
Stage 2: Construction Validation
- Prototype assembly review
- Fit and comfort evaluation
- Construction method verification
- Dimensional accuracy check
Stage 3: Performance Testing
- Durability test execution
- Environmental stress testing
- Safety compliance verification
- Quality parameter measurement
Stage 4: Final Approval
- Comprehensive results review
- Cost-benefit analysis
- Production readiness assessment
- Documentation completion
Each stage requires documented approval before progression, with clear criteria for advancement or rejection. This framework prevents costly production commitments on substandard samples.
Risk Assessment Matrix
Sample validation incorporates risk assessment methodologies that evaluate potential quality issues, market acceptance risks, and production challenges. This systematic approach prioritizes validation efforts and resource allocation.
| Risk Category | High Impact | Medium Impact | Low Impact |
|---|---|---|---|
| High Probability | Stop Production | Enhanced Testing | Standard Process |
| Medium Probability | Enhanced Testing | Standard Process | Reduced Testing |
| Low Probability | Standard Process | Reduced Testing | Minimal Testing |
Risk factors include material variability, construction complexity, regulatory requirements, and market expectations. Assessment results guide validation intensity and resource allocation decisions.
Supplier Performance Integration
Validation frameworks incorporate supplier performance history, capability assessments, and continuous improvement metrics. This integration ensures validation efforts align with supplier capabilities and development needs.
Supplier Evaluation Criteria:
- Historical quality performance
- Technical capability assessment
- Process control maturity
- Continuous improvement participation
- Compliance record maintenance
Performance data integration enables risk-based validation approaches, with enhanced scrutiny for new suppliers and streamlined processes for proven partners.
Validation Documentation and Compliance Requirements
Regulatory Compliance Frameworks
Sample validation must address regulatory requirements for target markets, including safety standards, chemical restrictions, and labeling requirements. Compliance documentation provides evidence of conformity and supports market access approvals.
Key Regulatory Standards:
- CE Marking (EN ISO 20347): European safety footwear requirements
- REACH Regulation: Chemical substance restrictions
- CPSIA: Consumer Product Safety Improvement Act (US children’s products)
- RoHS Directive: Restriction of hazardous substances
- OEKO-TEX Standards: Textile and leather ecological testing
[Shoe Manufacturer Guide](https://the manufacturer.com/shoe-manufacturer/)
Compliance validation requires accredited testing laboratories, documented test procedures, and certified results that support regulatory submissions and market approvals.
Quality Documentation Systems
Comprehensive documentation systems capture validation results, approval decisions, and corrective actions throughout the sample validation process. Digital documentation platforms enable efficient data management and retrieval for audits and reviews.
Documentation Requirements:
- Test Protocols: Standardized procedures and acceptance criteria
- Results Records: Quantitative data and photographic evidence
- Approval Certificates: Formal sign-offs and authorization records
- Corrective Actions: Issue identification and resolution tracking
- Supplier Communications: Feedback and improvement requirements
Documentation retention policies ensure availability for regulatory audits, customer inquiries, and continuous improvement initiatives.
Traceability and Chain of Custody
Sample validation establishes traceability systems that track materials, components, and processes throughout the validation cycle. This traceability supports quality investigations, supplier accountability, and regulatory compliance demonstrations.
Traceability Elements:
- Material Lot Numbers: Leather batch identification and tracking
- Component Serial Numbers: Hardware and accessory traceability
- Process Records: Manufacturing step documentation and operator identification
- Test Sample Identification: Unique identifiers linking samples to results
- Approval Timestamps: Decision points and authorization records
Chain of custody procedures ensure sample integrity and prevent contamination or substitution during validation processes.
Sample Validation Checklist for B2B Buyers
Pre-Validation Preparation Checklist
Material Specifications:
- Leather grade and quality requirements defined
- Color standards and tolerance ranges established
- Thickness specifications and measurement points identified
- Chemical compliance requirements documented
- Supplier certifications verified and current
Design and Construction:
- Technical drawings and specifications finalized
- Construction method selected and documented
- Size range and grading requirements established
- Fit requirements and testing protocols defined
- Quality standards and acceptance criteria documented
Testing Requirements:
- Applicable safety standards identified
- Durability testing protocols selected
- Environmental testing requirements established
- Performance benchmarks and targets defined
- Testing laboratory capabilities verified
Validation Execution Checklist
Sample Receipt and Initial Inspection:
- Sample quantity and identification verified
- Visual inspection completed and documented
- Dimensional measurements recorded
- Construction method verification performed
- Initial fit assessment conducted
Material Testing:
- Leather quality assessment completed
- Chemical composition testing performed
- Physical property measurements recorded
- Color fastness testing executed
- Defect identification and classification completed
Performance Validation:
- Durability testing initiated and monitored
- Environmental stress testing completed
- Safety compliance testing performed
- Comfort and fit validation conducted
- Statistical analysis of results completed
Post-Validation Decision Checklist
Results Analysis:
- All test results compiled and reviewed
- Compliance with specifications verified
- Risk assessment completed
- Cost-benefit analysis performed
- Supplier performance evaluation updated
Approval Process:
- Stakeholder review meetings conducted
- Approval criteria assessment completed
- Documentation package finalized
- Formal approval or rejection decision made
- Communication to supplier completed
Production Readiness:
- Production specifications finalized
- Quality control procedures established
- Supplier capability confirmation obtained
- Timeline and delivery schedules confirmed
- Contract terms and conditions agreed
[Shoe Manufacturer Guide](https://the manufacturer.com/shoe-manufacturer/)
This comprehensive checklist ensures systematic validation execution and reduces the risk of overlooked critical elements that could impact production quality or market acceptance.
Frequently Asked Questions
Q: How long does a complete leather shoe sample validation process typically take?
A: A comprehensive leather shoe sample validation process typically requires 3-6 weeks, depending on testing complexity and laboratory schedules. Material testing and basic quality assessments can be completed within 1-2 weeks, while durability testing requiring 100,000+ flex cycles may need 2-3 weeks. Environmental stress testing adds another 1-2 weeks for temperature cycling and chemical exposure protocols. Rush testing services are available but may compromise thoroughness and increase costs by 25-50%. Planning validation timelines early in the development process prevents delays and ensures adequate time for corrective actions if samples fail initial testing.
Q: What are the most critical quality parameters that cause sample rejection in leather shoe validation?
A: The top five critical parameters causing sample rejection include: 1) Dimensional inaccuracy exceeding ±3mm tolerance in length or width measurements, 2) Leather defects affecting more than 5% of visible surface area, 3) Construction failures including sole separation or stitching defects, 4) Chemical compliance failures particularly chromium VI levels exceeding 3mg/kg, and 5) Durability test failures including premature cracking or excessive wear after 50,000 flex cycles. These parameters directly impact product safety, performance, and marketability. Addressing these issues requires close collaboration with suppliers on material selection, process control, and quality assurance protocols.
Q: How do validation requirements differ between different target markets (US, EU, Asia)?
A: Validation requirements vary significantly across markets due to different regulatory frameworks and consumer expectations. European markets require CE marking compliance with EN ISO 20347 safety standards and REACH chemical regulations, emphasizing environmental sustainability and worker protection. US markets focus on CPSIA compliance for children’s footwear and slip resistance standards for workplace applications. Asian markets often emphasize durability testing and size accuracy due to different foot morphology and wear patterns. Chemical restrictions vary widely, with EU having the strictest limits on chromium VI (3mg/kg) while some Asian markets allow higher levels. Documentation requirements also differ, with EU requiring detailed technical files and US emphasizing test certificates from accredited laboratories.
Q: What role does statistical sampling play in leather shoe validation, and how many samples are needed?
A: Statistical sampling ensures validation results represent production quality rather than isolated sample performance. Sample size depends on production volume and quality risk levels, typically requiring 5-10 samples for initial validation and 3-5 samples for routine approvals. For high-volume production exceeding 10,000 pairs annually, statistical sampling plans following MIL-STD-105 or ISO 2859 standards determine appropriate sample sizes and acceptance criteria. Destructive testing requires additional samples since tested units cannot be used for further evaluation. Non-destructive testing like dimensional measurement can use smaller sample sizes. Risk-based sampling adjusts quantities based on supplier history, with new suppliers requiring larger sample sizes and proven suppliers qualifying for reduced sampling.
Q: How can B2B buyers optimize validation costs while maintaining quality standards?
A: Cost optimization strategies include: 1) Implementing risk-based testing that focuses resources on high-risk parameters while reducing testing frequency for proven stable characteristics, 2) Negotiating package deals with testing laboratories for multiple parameter testing, reducing per-test costs by 15-25%, 3) Utilizing supplier pre-qualification programs that verify capabilities before sample submission, reducing rejection rates and repeat testing, 4) Establishing preferred supplier relationships with validated quality systems, enabling streamlined validation processes, and 5) Investing in basic testing equipment for initial screening, reserving certified laboratory testing for final approval. Digital documentation systems reduce administrative costs and improve efficiency. Collaborative relationships with suppliers sharing validation costs and responsibilities create mutual incentives for quality improvement while reducing individual buyer expenses.
Ready to Start Your Shoe Line?
Transform your footwear concepts into market-ready products with professional sample validation services and manufacturing expertise. Our comprehensive quality assurance protocols ensure your leather shoes meet international standards while maintaining competitive pricing and reliable delivery schedules.
Contact our technical team to discuss your sample validation requirements and manufacturing needs. With over 17 years of experience and proven quality systems, we’re ready to support your success in the global footwear market.
Get Started Today:
- Email: peterwang@shwincheer.com
- Website: wincheershoes.com
- Minimum Order: 60 pairs for custom designs, 0 MOQ on existing styles